Class 11 Physics
Thermodynamics

First Law of Thermodynamics
- The First law of thermodynamics is same as law of conservation of energy.
- According to law of energy conservation: - Energy can neither be created nor be destroyed, only transformed to other forms.
- According to first law of thermodynamics:- The change in the internal energy of a closed system is equal to the amount of heat supplied to the system, minus the amount of work done by the system on its surroundings.
- Examples:- Consider a ball falling from the roof of the building when at top of the building the ball has only potential energy and when it starts falling potential energy decreases and kinetic energy starts increasing. At the ground it has only kinetic energy.
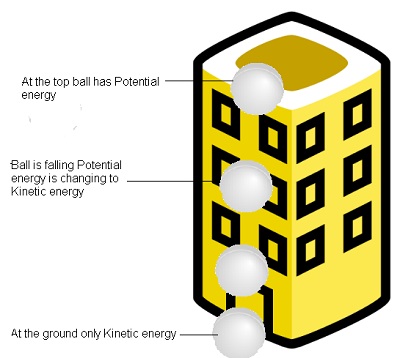
A ball falling from the roof of the building
Mathematically:-
ΔQ = ΔU + ΔW
Where:
- ΔQ is the heat supplied to the system by the surroundings
- ΔW is the work done by the system by the surroundings
- ΔU is the change in internal energy of the system
- Some part of heat supplied gets lost and remaining part is the work done on the surroundings. This remaining part is used up to increase or change the internal energy of the system.
ΔQ = ΔU + ΔW
Consider a system whose initial state is (P1, V1) and final state (P2, V2)
ΔU is the change in the energy of the system to change from initial state to final state.
- Internal energy is a state variable which means it is path independent. It does not depend how state changes from initial to final.
- But the work done and heat is path dependent. It depends on how the path changes from initial to final.
- Consider a system whose initial state is defined as (P1,V1) and Final state is defined as (P2,V2).
- The internal energy doesn’t depend on how the system has changed from initial state to final state. It only depends on how it has reached from initial state to final state.
Therefore:-
ΔQ – ΔW = ΔU where
- (ΔQ and ΔW are path dependent quantities whereas ΔU is path independent quantity)
- This concludes ΔQ – ΔW is path independent quantity.
- Case 1:- System undergoes a process such that ΔU = 0 which means internal energy is constant. From first law of thermodynamics
ΔQ = ΔU + ΔW putting ΔU = 0
- This implies ΔQ = ΔW this means heat supplied by the surroundings is equal to the work done by the system on the surroundings.
- Case 2:- System is a gas in a cylinder with movable piston, by moving the piston we can change the volume of the gas.
- If we move the piston downwards some work is done and it can be given as:-
- Work done = ΔW
- = Force x displacement
- = P x Area x displacement
- ΔW = PΔV (ΔV= Area x displacement) (Equation 1)
- Therefore by first law of thermodynamics
- ΔQ = ΔU + PΔV where ΔV= change in volume
(From Equation 1)
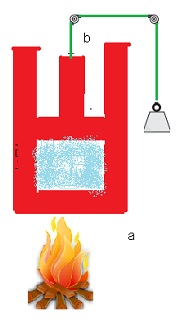
Heat and work are two distinct modes of energy transfer to a system that results in change in its internal energy. (a) Heat is energy transfer due to temperature difference between the system and the surroundings. (b) Work is energy transfer brought about by means (e.g. moving the piston by raising or lowering some weight connected to it) that do not involve such a temperature difference.
Problem: An electric heater supplies heat to a system at a rate of 100W. If system performs work at a rate of 75 Joules per second. At what rate is the internal energy increasing?
Answer:
Heat is supplied to the system at a rate of 100 W.
Heat supplied, Q = 100 J/s
The system performs at a rate of 75 J/s.
Work done, W = 75 J/s
From the first law of thermodynamics, we have:
Q = U + W
Where, U = Internal energy
U = Q – W
= 100 – 75
= 25 J/s
= 25 W
Therefore, the internal energy of the given electric heater increases at a rate of 25 W.
Problem: A cylinder filled with gas and fitted with a movable piston. In changing the state of a gas adiabatically from equilibrium state A to another equilibrium state B, an amount of work equal to 22.3 J is done on the system. If the gas is taken from state A to B via a process in which the net heat absorbed by the system is 9.35 cal, how much is the net work done by the system in the latter case? (Take 1 cal = 4.19 J)
Answer:
The work done (W) on the system while the gas changes from state A to state B is 22.3 J.
This is an adiabatic process. Hence, change in heat is zero.
ΔQ = 0
ΔW = –22.3 J (Since the work is done on the system)
From the first law of thermodynamics, we have:
ΔQ = ΔU + ΔW
Where,
ΔU = Change in the internal energy of the gas
ΔU = ΔQ – ΔW = – (– 22.3 J)
ΔU = + 22.3 J
When the gas goes from state A to state B via a process, the net heat absorbed by the system is:
ΔQ = 9.35 cal = 9.35 × 4.19 = 39.1765 J
Heat absorbed, ΔQ = ΔU + ΔQ
ΔW = ΔQ – ΔU
= 39.1765 – 22.3
= 16.8765 J
Therefore, 16.88 J of work is done by the system.
Problem: Two cylinders A and B of equal capacity are connected to each other via a stopcock. A contains a gas at standard temperature and pressure. B is completely evacuated. The entire system is thermally insulated. The stopcock is suddenly opened. Answer the following:
(a) What is the final pressure of the gas in A and B?
(b) What is the change in internal energy of the gas?
(c) What is the change in the temperature of the gas?
(d) Do the intermediate states of the system (before settling to the final equilibrium state) lie on its P-V-T surface?
Answer:
(a) When the stopcock is suddenly opened, the volume available to the gas at 1 atmospheric pressure will become two times. Therefore, pressure will decrease to one-half, i.e., 0.5 atmosphere.
(b) There will be no change in the internal energy of the gas as no work is done on/by the gas.
(c) Since no work is being done by the gas during the expansion of the gas, the temperature of the gas will not change at all.
(d) No, because the process called free expansion is rapid and cannot be controlled. the intermediate states are non-equilibrium states and do not satisfy the gas equation. In due course, the gas does return to an equilibrium state.
Class 11 Physics Thermodynamics NCERT Chapter 11 Free Notes for Best Revision
Revision of Class 11 Physics Thermodynamics is a crucial aspect of effective learning. Revision plays a vital role in the learning process and is especially important before exams. Here are some key points you can consider emphasizing in your content:
- Retention and Memory: Regularly reviewing and revisiting the material of Class 11 Physics Thermodynamics helps reinforce the concepts in students' minds. It strengthens memory pathways, making it easier to recall information during exams and beyond.
- Consolidation of Knowledge: When you revise notes, you are essentially consolidating your knowledge. This means connecting new information with what you already know, making the overall understanding more robust.
- Identifying Knowledge Gaps: Revision allows students to identify any gaps in their understanding or areas where they need further clarification. This gives you a chance to seek help or delve deeper into those topics. For detailed understanding, you can always refer to the videos of Thermodynamics Class 11 Physics NCERT Chapter 11 on LearnoHub.com
- Building Confidence: As you revise Thermodynamics Class 11 Physics and become more familiar with the content, your confidence in your abilities grows. Confidence is a crucial factor in exam performance as it reduces anxiety and allows you to approach exams with a positive mindset.
- Different Revision Techniques: Use a variety of revision techniques such as summarizing notes, creating flashcards, practicing past papers, discussing concepts with peers, and teaching others. Different techniques work for different students, and it's essential to find what suits you the best. You can also attend the LIVE Revision classes on LearnoHub.com or watch the LIVE Revision Race videos of LearnoHub on Thermodynamics Class 11 Physics NCERT Chapter 11.
- Spacing Effect: Spacing out revision sessions over time, rather than cramming all at once, has been shown to improve long-term retention. Create a revision schedule leading up to the exams to allow for spaced practice.
- Regular Revision over Cramming: Regular and consistent revision throughout the academic year is very important. Waiting until the last moment to cram everything can be overwhelming and less effective than spaced-out revision.
- Self-Assessment: Assess your understanding periodically through quizzes or self-tests. This helps you to gauge your progress and identify areas that need further attention. Refer Class 11 Thermodynamics Online Tests.
- Balanced Approach: Remind students to strike a balance between revision and other activities. Adequate rest, exercise, and relaxation are essential for optimal learning and performance.
- Seeking Help: If you face difficulties during the revision process, Refer the videos of Class 11 Physics Thermodynamics. Clearing doubts promptly is crucial for a better grasp of the subject matter. You can always ask your doubts on Thermodynamics Class 11 Physics NCERT Chapter 11. “Ask a Question” section of LearnoHub.com
By highlighting the benefits and strategies of effective revision, you can approach your studies more mindfully and achieve better results in your exams. Best of luck bachhon!
Class 11 Physics seems to be a quite difficult subject for a lot of students. But, if you get a very good conceptual understanding of the subject, it can be very interesting for you.
We, at LearnoHub, will give our best to make Class 11 Physics Thermodynamics NCERT Chapter 11 super-duper easy for you.
We aim at making learning fun as well as engaging for you with our complete end-end learning content with Thermodynamics Class 11 Physics Best videos, Notes, NCERT pdf, NCERT complete syllabus, tests and Practice Questions.
Always remember, it is very important to study with full concentration during Revision. Here are a few tips for you on how to revise with full focus:
- Create a Distraction-Free Environment: Find a quiet and comfortable place to study where you can minimize distractions. Turn off or silence your phone, log out of social media accounts, and inform others around you that you need uninterrupted study time. A dedicated study environment will help you focus better.
- Set Specific Goals: Before starting your study session, set clear and achievable goals. Break down your study material into smaller tasks, and plan what you want to accomplish during each session. Having specific goals will give you a sense of direction and purpose, making it easier to concentrate.
- Use the Pomodoro Technique: The Pomodoro Technique is a time management method that involves studying in short, focused intervals, typically 25 minutes, followed by a short break of 5 minutes. After completing four sessions, take a longer break of around 15-30 minutes. This technique can improve focus and productivity by providing regular breaks to recharge.
- Stay Organized: Keep your study materials, notes, and resources well-organized. Having everything you need at hand will save time and reduce distractions caused by searching for materials. Use color-coded folders or digital tools to maintain a structured study system.
- Practice Mindfulness and Meditation: Before you begin studying, take a few minutes to practice mindfulness or meditation. Deep breathing exercises and clearing your mind of distractions can help you approach your study session with a calm and focused mindset.
Remember, studying with full concentration is a skill that takes time and practice to develop. If you find your mind wandering during study sessions, gently bring your focus back to the task at hand and be patient with yourself. With consistent effort, you can improve your ability to concentrate and make the most of your study time.
Last but not the least, To get the best hold on Class 11 Physics Thermodynamics Book Chapter 11. Do not forget to check out:
- Thermodynamics Class 11 Physics Best videos
- Thermodynamics Class 11 Physics NCERT Solutions
- Class 11 Physics Thermodynamics Revision notes
- Thermodynamics Class 11 Physics DPPS, Download PDF of solutions
- Class 11 Physics Thermodynamics Online Tests
- Class 11 Physics Sample papers
