

Question:
Give one chemical test to distinguish between the following pairs of compounds. (i) Methylamine and dimethylamine (ii) Secondary and tertiary amines (iii) Ethylamine and aniline (iv) Aniline and benzylamine (v) Aniline and N-methylaniline.
Answer:
(i) Methylamine and dimethylamine can be distinguished by the carbylamine test.
Carbylamine test: Aliphatic and aromatic primary amines on heating with chloroform and ethanolic potassium hydroxide form foul-smelling isocyanides or carbylamines. Methylamine (being an aliphatic primary amine) gives a positive carbylamine test, but dimethylamine does not.
(ii) Secondary and tertiary amines can be distinguished by allowing them to react with Hinsberg’s reagent (benzenesulphonyl chloride, 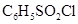 ).
).
Secondary amines react with Hinsberg’s reagent to form a product that is insoluble in an alkali. For example, N, N – diethylamine reacts with Hinsberg’s reagent to form N, N – diethylbenzenesulphonamide, which is insoluble in an alkali. Tertiary amines, however, do not react with Hinsberg’s reagent.
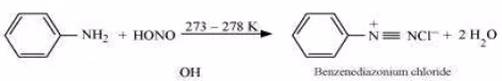
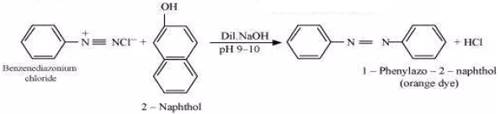
(iii) Ethylamine and aniline can be distinguished using the azo-dye test. A dye is obtained when aromatic amines react with 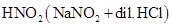 at
at  , followed by a reaction with the alkaline solution of 2-naphthol. The dye is usually yellow, red, or orange in colour. Aliphatic amines give a brisk effervescence due (to the evolution of
, followed by a reaction with the alkaline solution of 2-naphthol. The dye is usually yellow, red, or orange in colour. Aliphatic amines give a brisk effervescence due (to the evolution of  gas) under similar conditions.
gas) under similar conditions.
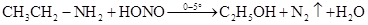
(iv) Aniline and benzylamine can be distinguished by their reactions with the help of nitrous acid, which is prepared in situ from a mineral acid and sodium nitrite. Benzylamine reacts with nitrous acid to form unstable diazonium salt, which in turn gives alcohol with the evolution of nitrogen gas.
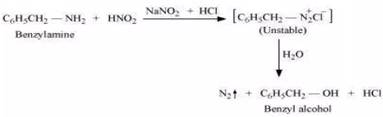
On the other hand, aniline reacts with 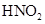 at a low temperature to form stable diazonium salt. Thus, nitrogen gas is not evolved.
at a low temperature to form stable diazonium salt. Thus, nitrogen gas is not evolved.
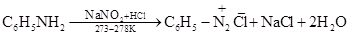
(v) Aniline and N-methylaniline can be distinguished using the Carbylamine test. Primary amines, on heating with chloroform and ethanolic potassium hydroxide, form foul-smelling isocyanides or carbylamines. Aniline, being an aromatic primary amine, gives positive carbylamine test. However, N-methylaniline, being a secondary amine does not.
Not what you are looking for? Go ahead and submit the question, we will get back to you.
