

Question:
sir i am unable to get the orbital concept of hyperconjuation
Answer:
Hyperconjugation is the stabilising interaction that results from the interaction of the electrons in a s-bond (usually C-H or C-C) with an adjacent empty (or partially filled) p-orbital or a p-orbital to give an extended molecular orbital that increases the stability of the system.
| Lets consider how a methyl group is involved in hyperconjugation with a carbocation centre. | 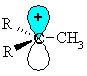 |
| First we need to draw it to show the C-H s-bonds. Note that the empty p orbital associated with the positive charge at the carbocation centre is in the same plane (i.e. coplanar) with one of the C-H s-bonds (shown in blue.) |
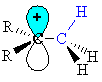 |
|
This geometry means the electrons in the s-bond can be stabilised by an interaction with the empty p-orbital of the carbocation centre.
|
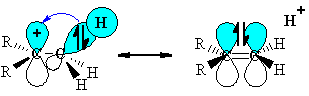 |
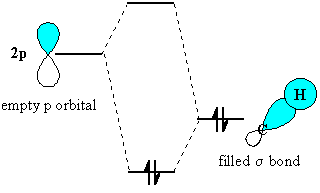
The stabilisation arises because the orbital interaction leads to the electrons being in a lower energy orbital
Not what you are looking for? Go ahead and submit the question, we will get back to you.
